Mathematical and theoretical biology
Mathematical and theoretical biology is an interdisciplinary scientific research field with a range of applications in biology, biotechnology, and medicine.[1] The field is also called mathematical biology or biomathematics to stress the mathematical side, or theoretical biology to stress the biological side.[2] Mathematical biology aims at the mathematical representation, treatment and modeling of biological processes, using a variety of applied mathematical techniques and tools. It has both theoretical and practical applications in biological, biomedical and biotechnology research. For example, in cell biology, protein interactions are often represented as "cartoon" models, which, although easy to visualize, do not accurately describe the systems studied. This requires precise mathematical models. Describing systems in a quantitative manner means their behavior can be better simulated, and hence properties can be predicted that might not be evident to the experimenter.
Mathematical biology may employ calculus, probability theory, statistics, linear algebra, abstract algebra, graph theory, combinatorics, algebraic geometry, topology, dynamical systems, differential equations and coding theory.[3] Some mathematical areas, such as certain methodologies in statistics, were developed as tools during the conduct of research into mathematical biology.
Importance
Applying mathematics to biology has a long history, but only recently has there been an explosion of interest in the field. Some reasons for this include:
- The explosion of data-rich information sets, due to the genomics revolution, which are difficult to understand without the use of analytical tools
- Recent development of mathematical tools such as chaos theory to help understand complex, non-linear mechanisms in biology
- An increase in computing power, which facilitates calculations and simulations not previously possible
- An increasing interest in in silico experimentation due to ethical considerations, risk, unreliability and other complications involved in human and animal research
Areas of research
Several areas of specialized research in mathematical and theoretical biology[4][5][6][7][8] as well as external links to related projects in various universities are concisely presented in the following subsections, including also a large number of appropriate validating references from a list of several thousands of published authors contributing to this field. Many of the included examples are characterised by highly complex, nonlinear, and supercomplex mechanisms, as it is being increasingly recognised that the result of such interactions may only be understood through a combination of mathematical, logical, physical/chemical, molecular and computational models. Due to the wide diversity of specific knowledge involved, biomathematical research is often done in collaboration between mathematicians, biomathematicians, theoretical biologists, physicists, biophysicists, biochemists, bioengineers, engineers, biologists, physiologists, research physicians, biomedical researchers, oncologists, molecular biologists, geneticists, embryologists, zoologists, chemists, etc.
Evolutionary biology
Ecology and evolutionary biology have traditionally been the dominant fields of mathematical biology.
Evolutionary biology has been the subject of extensive mathematical theorizing. The traditional approach in this area, which includes complications from genetics, is population genetics. Most population geneticists consider the appearance of new alleles by mutation, the appearance of new genotypes by recombination, and changes in the frequencies of existing alleles and genotypes at a small number of gene loci. When infinitesimal effects at a large number of gene loci are considered, together with the assumption of linkage equilibrium or quasi-linkage equilibrium, one derives quantitative genetics. Ronald Fisher made fundamental advances in statistics, such as analysis of variance, via his work on quantitative genetics. Another important branch of population genetics that led to the extensive development of coalescent theory is phylogenetics. Phylogenetics is an area that deals with the reconstruction and analysis of phylogenetic (evolutionary) trees and networks based on inherited characteristics[9] Traditional population genetic models deal with alleles and genotypes, and are frequently stochastic.
Many population genetics models assume that population sizes are constant. Variable population sizes, often in the absence of genetic variation, are treated by the field of population dynamics. Work in this area dates back to the 19th century, and even as far as 1798 when Thomas Malthus formulated the first principle of population dynamics, which later became known as the Malthusian growth model. The Lotka–Volterra predator-prey equations are another famous example. Population dynamics overlap with another active area of research in mathematical biology: mathematical epidemiology, the study of infectious disease affecting populations. Various models of the spread of infections have been proposed and analyzed, and provide important results that may be applied to health policy decisions.
In evolutionary game theory, developed first by John Maynard Smith and George R. Price, selection acts directly on inherited phenotypes, without genetic complications. This approach has been mathematically refined to produce the field of adaptive dynamics.
Computer models and automata theory
A monograph on this topic summarizes an extensive amount of published research in this area up to 1986,[10][11][12] including subsections in the following areas: computer modeling in biology and medicine, arterial system models, neuron models, biochemical and oscillation networks, quantum automata, quantum computers in molecular biology and genetics,[13] cancer modelling,[14] neural nets, genetic networks, abstract categories in relational biology,[15] metabolic-replication systems, category theory[16] applications in biology and medicine,[17] automata theory, cellular automata,[18] tessellation models[19][20] and complete self-reproduction, chaotic systems in organisms, relational biology and organismic theories.[21][22] This published report also includes 390 references to peer-reviewed articles by a large number of authors.[4][23][24]
Modeling cell and molecular biology
This area has received a boost due to the growing importance of molecular biology.[7]
- Mechanics of biological tissues[25]
- Theoretical enzymology and enzyme kinetics
- Cancer modelling and simulation[26][27]
- Modelling the movement of interacting cell populations[28]
- Mathematical modelling of scar tissue formation[29]
- Mathematical modelling of intracellular dynamics[30][31]
- Mathematical modelling of the cell cycle[32]
Modelling physiological systems
Molecular set theory
Molecular set theory (MST) is a mathematical formulation of the wide-sense chemical kinetics of biomolecular reactions in terms of sets of molecules and their chemical transformations represented by set-theoretical mappings between molecular sets. It was introduced by Anthony Bartholomay, and its applications were developed in mathematical biology and especially in mathematical medicine.[35] In a more general sense, MST is the theory of molecular categories defined as categories of molecular sets and their chemical transformations represented as set-theoretical mappings of molecular sets. The theory has also contributed to biostatistics and the formulation of clinical biochemistry problems in mathematical formulations of pathological, biochemical changes of interest to Physiology, Clinical Biochemistry and Medicine.[35][36]
Complex systems biology
An elaboration of systems biology to understanding the more complex life processes was developed since 1970 in connection with molecular set theory, relational biology and algebraic biology.
Mathematical methods
A model of a biological system is converted into a system of equations, although the word 'model' is often used synonymously with the system of corresponding equations. The solution of the equations, by either analytical or numerical means, describes how the biological system behaves either over time or at equilibrium. There are many different types of equations and the type of behavior that can occur is dependent on both the model and the equations used. The model often makes assumptions about the system. The equations may also make assumptions about the nature of what may occur.
Simulation of mathematical biology
Computer with significant recent evolution in performance acceraretes the model simulation based on various formulas. The websites BioMath Modeler can run simulations and display charts interactively on browser.
Mathematical biophysics
The earlier stages of mathematical biology were dominated by mathematical biophysics, described as the application of mathematics in biophysics, often involving specific physical/mathematical models of biosystems and their components or compartments.
The following is a list of mathematical descriptions and their assumptions.
Deterministic processes (dynamical systems)
A fixed mapping between an initial state and a final state. Starting from an initial condition and moving forward in time, a deterministic process always generates the same trajectory, and no two trajectories cross in state space.
- Difference equations/Maps – discrete time, continuous state space.
- Ordinary differential equations – continuous time, continuous state space, no spatial derivatives. See also: Numerical ordinary differential equations.
- Partial differential equations – continuous time, continuous state space, spatial derivatives. See also: Numerical partial differential equations.
- Logical deterministic cellular automata – discrete time, discrete state space. See also: Cellular automaton.
Stochastic processes (random dynamical systems)
A random mapping between an initial state and a final state, making the state of the system a random variable with a corresponding probability distribution.
- Non-Markovian processes – generalized master equation – continuous time with memory of past events, discrete state space, waiting times of events (or transitions between states) discretely occur.
- Jump Markov process – master equation – continuous time with no memory of past events, discrete state space, waiting times between events discretely occur and are exponentially distributed. See also: Monte Carlo method for numerical simulation methods, specifically dynamic Monte Carlo method and Gillespie algorithm.
- Continuous Markov process – stochastic differential equations or a Fokker-Planck equation – continuous time, continuous state space, events occur continuously according to a random Wiener process.
Spatial modelling
One classic work in this area is Alan Turing's paper on morphogenesis entitled The Chemical Basis of Morphogenesis, published in 1952 in the Philosophical Transactions of the Royal Society.
- Travelling waves in a wound-healing assay[37]
- Swarming behaviour[38]
- A mechanochemical theory of morphogenesis[39]
- Biological pattern formation[40]
- Spatial distribution modeling using plot samples[41]
Relational biology
Abstract relational biology (ARB)[42] is concerned with the study of general, relational models of complex biological systems, usually abstracting out specific morphological, or anatomical, structures. Some of the simplest models in ARB are the Metabolic-Replication, or (M,R)--systems introduced by Robert Rosen in 1957-1958 as abstract, relational models of cellular and organismal organization.
Algebraic biology
Algebraic biology (also known as symbolic systems biology) applies the algebraic methods of symbolic computation to the study of biological problems, especially in genomics, proteomics, analysis of molecular structures and study of genes.[21][43][44]
Model example: the cell cycle
The eukaryotic cell cycle is very complex and is one of the most studied topics, since its misregulation leads to cancers. It is possibly a good example of a mathematical model as it deals with simple calculus but gives valid results. Two research groups [45][46] have produced several models of the cell cycle simulating several organisms. They have recently produced a generic eukaryotic cell cycle model that can represent a particular eukaryote depending on the values of the parameters, demonstrating that the idiosyncrasies of the individual cell cycles are due to different protein concentrations and affinities, while the underlying mechanisms are conserved (Csikasz-Nagy et al., 2006).
By means of a system of ordinary differential equations these models show the change in time (dynamical system) of the protein inside a single typical cell; this type of model is called a deterministic process (whereas a model describing a statistical distribution of protein concentrations in a population of cells is called a stochastic process).
To obtain these equations an iterative series of steps must be done: first the several models and observations are combined to form a consensus diagram and the appropriate kinetic laws are chosen to write the differential equations, such as rate kinetics for stoichiometric reactions, Michaelis-Menten kinetics for enzyme substrate reactions and Goldbeter–Koshland kinetics for ultrasensitive transcription factors, afterwards the parameters of the equations (rate constants, enzyme efficiency coefficients and Michaelis constants) must be fitted to match observations; when they cannot be fitted the kinetic equation is revised and when that is not possible the wiring diagram is modified. The parameters are fitted and validated using observations of both wild type and mutants, such as protein half-life and cell size.
To fit the parameters, the differential equations must be studied. This can be done either by simulation or by analysis. In a simulation, given a starting vector (list of the values of the variables), the progression of the system is calculated by solving the equations at each time-frame in small increments.
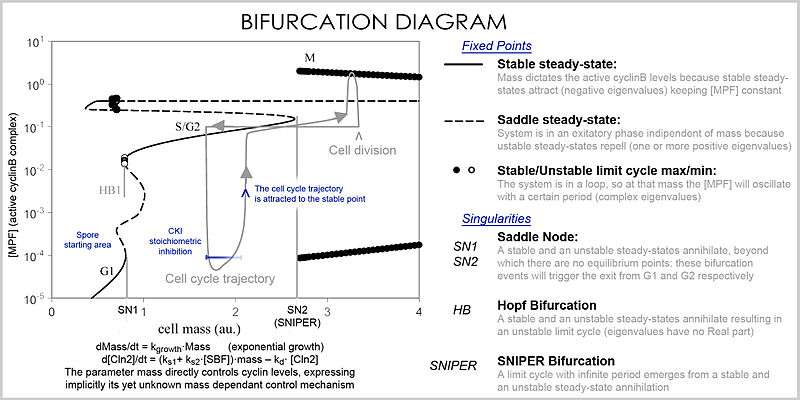
In analysis, the properties of the equations are used to investigate the behavior of the system depending of the values of the parameters and variables. A system of differential equations can be represented as a vector field, where each vector described the change (in concentration of two or more protein) determining where and how fast the trajectory (simulation) is heading. Vector fields can have several special points: a stable point, called a sink, that attracts in all directions (forcing the concentrations to be at a certain value), an unstable point, either a source or a saddle point, which repels (forcing the concentrations to change away from a certain value), and a limit cycle, a closed trajectory towards which several trajectories spiral towards (making the concentrations oscillate).
A better representation, which handles the large number of variables and parameters, is a bifurcation diagram using bifurcation theory. The presence of these special steady-state points at certain values of a parameter (e.g. mass) is represented by a point and once the parameter passes a certain value, a qualitative change occurs, called a bifurcation, in which the nature of the space changes, with profound consequences for the protein concentrations: the cell cycle has phases (partially corresponding to G1 and G2) in which mass, via a stable point, controls cyclin levels, and phases (S and M phases) in which the concentrations change independently, but once the phase has changed at a bifurcation event (Cell cycle checkpoint), the system cannot go back to the previous levels since at the current mass the vector field is profoundly different and the mass cannot be reversed back through the bifurcation event, making a checkpoint irreversible. In particular the S and M checkpoints are regulated by means of special bifurcations called a Hopf bifurcation and an infinite period bifurcation.
See also
- Artificial life
- Bio-inspired computing
- Biological applications of bifurcation theory
- Biosemiotics
- Biostatistics
- Cellular automaton
- Computational biology
- Computational gene
- DNA computing
- Entropy and life
- Ewens's sampling formula
- Journal of Theoretical Biology
- Mathematical modelling of infectious disease
- Metabolic network modelling
- Molecular modelling
- Morphometrics
- Population genetics
- Protein folding
- Simulated reality
- Statistical genetics
- Systems biology
- Theoretical ecology
- Quantum Biology
Societies and institutes
- National Institute for Mathematical and Biological Synthesis
- Society for Mathematical Biology
- ESMTB: European Society for Mathematical and Theoretical Biology
- The Israeli Society for Theoretical and Mathematical Biology
- Société Francophone de Biologie Théorique
- International Society for Biosemiotic Studies
Notes
- ↑ Mathematical and Theoretical Biology: A European Perspective
- ↑ "There is a subtle difference between mathematical biologists and theoretical biologists. Mathematical biologists tend to be employed in mathematical departments and to be a bit more interested in math inspired by biology than in the biological problems themselves, and vice versa." Careers in theoretical biology
- ↑ Robeva, Raina; et al. (Fall 2010). "Mathematical Biology Modules Based on Modern Molecular Biology and Modern Discrete Mathematics". CBE Life Sciences Education. The American Society for Cell Biology. 9 (3): 227–240. doi:10.1187/cbe.10-03-0019. PMC 2931670
 . PMID 20810955.
. PMID 20810955. - 1 2 Baianu, I. C.; Brown, R.; Georgescu, G.; Glazebrook, J. F. (2006). "Complex Non-linear Biodynamics in Categories, Higher Dimensional Algebra and Łukasiewicz–Moisil Topos: Transformations of Neuronal, Genetic and Neoplastic Networks". Axiomathes. 16: 65–122. doi:10.1007/s10516-005-3973-8.
- ↑ Łukasiewicz-Topos Models of Neural Networks, Cell Genome and Interactome Nonlinear Dynamic Models (2004) http://cogprints.org/3701/01/ANeuralGenNetworkLuknTopos_oknu4.pdf/
- ↑ Complex Systems Analysis of Arrested Neural Cell Differentiation during Development and Analogous Cell Cycling Models in Carcinogenesis (2004) http://cogprints.org/3687/
- 1 2 "Research in Mathematical Biology". Maths.gla.ac.uk. Retrieved 2008-09-10.
- ↑ J. R. Junck. Ten Equations that Changed Biology: Mathematics in Problem-Solving Biology Curricula, Bioscene, (1997), 23(1):11-36 New Link (Aug 2010)
- ↑ Charles Semple (2003), Phylogenetics, Oxford University Press, ISBN 978-0-19-850942-4
- ↑ "Computer Models and Automata Theory in Biology and Medicine" (1986). In:Mathematical Modeling: Mathematical Models in Medicine, volume 7:1513-1577, M. Witten, Ed., Pergamon Press: New York. http://cdsweb.cern.ch/record/746663/files/COMPUTER_MODEL_AND_AUTOMATA_THEORY_IN_BIOLOGY2p.pdf
- ↑ Lin, H.C. 2004. "Computer Simulations and the Question of Computability of Biological Systems": 1-15,doi=10.1.1.108.5072. https://tspace.library.utoronto.ca/bitstream/1807/2951/2/compauto.pdf
- ↑ "Computer Models and Automata Theory in Biology and Medicine" (1986). (Abstract) http://biblioteca.universia.net/html_bura/ficha/params/title/computer-models-and-automata-theory-in-biology-and-medicine/id/3920559.html
- ↑ "Natural Transformations Models in Molecular Biology"(1983). In: SIAM and Society of Mathematical Biology, National Meeting, Bethesda,MD:1-12. http://citeseerx.ist.psu.edu/showciting;jsessionid=BD12D600C39F9979633DB877CA74212B?cid=642862
- ↑ "Quantum Interactomics and Cancer Mechanisms" (2004): 1-16, Research Report communicated to the Institute of Genomic Biology, University of Illinois at Urbana https://tspace.library.utoronto.ca/retrieve/4969/QuantumInteractomicsInCancer_Sept13k4E_cuteprt.pdf
- ↑ Kainen,P.C. 2005."Category Theory and Living Systems", In: Charles Ehresmann's Centennial Conference Proceedings: 1-5,University of Amiens, France, October 7-9th, 2005, A. Ehresmann, Organizer and Editor. http://vbm-ehr.pagesperso-orange.fr/ChEh/articles/Kainen.pdf
- ↑ "bibliography for category theory/algebraic topology applications in physics". PlanetPhysics. Retrieved 2010-03-17.
- ↑ "bibliography for mathematical biophysics and mathematical medicine". PlanetPhysics. 2009-01-24. Retrieved 2010-03-17.
- ↑ "Cellular Automata". Los Alamos Science. Fall 1983.
- ↑ Modern Cellular Automata by Kendall Preston and M. J. B. Duff https://books.google.com/books?id=l0_0q_e-u_UC&dq=cellular+automata+and+tessalation&pg=PP1&ots=ciXYCF3AYm&source=citation&sig=CtaUDhisM7MalS7rZfXvp689y-8&hl=en&sa=X&oi=book_result&resnum=12&ct=result
- ↑ "Dual Tessellation – from Wolfram MathWorld". Mathworld.wolfram.com. 2010-03-03. Retrieved 2010-03-17.
- 1 2 Baianu, I. C. 1987, Computer Models and Automata Theory in Biology and Medicine., in M. Witten (ed.),Mathematical Models in Medicine, vol. 7., Ch.11 Pergamon Press, New York, 1513-1577. http://cogprints.org/3687/
- ↑ "Computer models and automata theory in biology and medicine | KLI Theory Lab". Theorylab.org. 2009-05-26. Retrieved 2010-03-17.
- ↑ Currently available for download as an updated PDF: http://cogprints.ecs.soton.ac.uk/archive/00003718/01/COMPUTER_SIMULATIONCOMPUTABILITYBIOSYSTEMSrefnew.pdf
- ↑ "bibliography for mathematical biophysics". PlanetPhysics. Retrieved 2010-03-17.
- ↑ Ray Ogden (2004-07-02). "rwo_research_details". Maths.gla.ac.uk. Retrieved 2010-03-17.
- ↑ Oprisan, Sorinel A.; Oprisan, Ana (2006). "A Computational Model of Oncogenesis using the Systemic Approach". Axiomathes. 16: 155–163. doi:10.1007/s10516-005-4943-x.
- ↑ "MCRTN – About tumour modelling project". Calvino.polito.it. Retrieved 2010-03-17.
- ↑ "Jonathan Sherratt's Research Interests". Ma.hw.ac.uk. Retrieved 2010-03-17.
- ↑ "Jonathan Sherratt's Research: Scar Formation". Ma.hw.ac.uk. Retrieved 2010-03-17.
- ↑ Kuznetsov, A.V.; Avramenko, A.A. (2009). "A macroscopic model of traffic jams in axons". Mathematical Biosciences. 218 (2): 142–152. doi:10.1016/j.mbs.2009.01.005.
- ↑ http://www.sbi.uni-rostock.de/dokumente/p_gilles_paper.pdf
- ↑ Archived July 28, 2007, at the Wayback Machine.
- ↑ Hassan Ugail. "Department of Mathematics – Prof N A Hill's Research Page". Maths.gla.ac.uk. Retrieved 2010-03-17.
- ↑ "Integrative Biology – Heart Modelling". Integrativebiology.ox.ac.uk. Retrieved 2010-03-17.
- 1 2 "molecular set category". PlanetPhysics. Retrieved 2010-03-17.
- ↑ Representation of Uni-molecular and Multimolecular Biochemical Reactions in terms of Molecular Set Transformations http://planetmath.org/?op=getobj&from=objects&id=10770[]
- ↑ "Travelling waves in a wound". Maths.ox.ac.uk. Retrieved 2010-03-17.
- ↑
- ↑ "The mechanochemical theory of morphogenesis". Maths.ox.ac.uk. Retrieved 2010-03-17.
- ↑ "Biological pattern formation". Maths.ox.ac.uk. Retrieved 2010-03-17.
- ↑ Hurlbert, Stuart H. (1990). "Spatial Distribution of the Montane Unicorn". Oikos. 58 (3): 257–271. doi:10.2307/3545216. JSTOR 3545216.
- ↑ Abstract Relational Biology (ARB)
- ↑ Michael P Barnett, "Symbolic calculation in the life sciences: trends and prospects, Algebraic Biology 2005" – Computer Algebra in Biology, edited by H. Anai, K. Horimoto, Universal Academy Press, Tokyo, 2006. (on line .pdf format)
- ↑ http://library.bjcancer.org/ebook/109.pdf L. Preziosi, Cancer Modelling and Simulation. Chapman Hall/CRC Press, 2003. ISBN 1-58488-361-8.
- ↑ "The JJ Tyson Lab". Virginia Tech. Retrieved 2008-09-10.
- ↑ "The Molecular Network Dynamics Research Group". Budapest University of Technology and Economics.
References
- D. Barnes; D. Chu (2010). Introduction to Modelling for Biosciences. Springer Verlag. ISBN 1-84996-325-8.
- Israel G (1988). "On the contribution of Volterra and Lotka to the development of modern biomathematics". History and Philosophy of the Life Sciences. 10 (1): 37–49. PMID 3045853.
- Scudo FM (March 1971). "Vito Volterra and theoretical ecology". Theoretical Population Biology. 2 (1): 1–23. doi:10.1016/0040-5809(71)90002-5. PMID 4950157.
- S.H. Strogatz, Nonlinear dynamics and Chaos: Applications to Physics, Biology, Chemistry, and Engineering. Perseus, 2001, ISBN 0-7382-0453-6
- N.G. van Kampen, Stochastic Processes in Physics and Chemistry, North Holland., 3rd ed. 2001, ISBN 0-444-89349-0
- I. C. Baianu., Computer Models and Automata Theory in Biology and Medicine., Monograph, Ch.11 in M. Witten (Editor), Mathematical Models in Medicine, vol. 7., Vol. 7: 1513-1577 (1987),Pergamon Press:New York, (updated by Hsiao Chen Lin in 2004 ISBN 0-08-036377-6
- P.G. Drazin, Nonlinear systems. C.U.P., 1992. ISBN 0-521-40668-4
- L. Edelstein-Keshet, Mathematical Models in Biology. SIAM, 2004. ISBN 0-07-554950-6
- G. Forgacs and S. A. Newman, Biological Physics of the Developing Embryo. C.U.P., 2005. ISBN 0-521-78337-2
- A. Goldbeter, Biochemical oscillations and cellular rhythms. C.U.P., 1996. ISBN 0-521-59946-6
- L.G. Harrison, Kinetic theory of living pattern. C.U.P., 1993. ISBN 0-521-30691-4
- F. Hoppensteadt, Mathematical theories of populations: demographics, genetics and epidemics. SIAM, Philadelphia, 1975 (reprinted 1993). ISBN 0-89871-017-0
- D.W. Jordan and P. Smith, Nonlinear ordinary differential equations, 2nd ed. O.U.P., 1987. ISBN 0-19-856562-3
- J.D. Murray, Mathematical Biology. Springer-Verlag, 3rd ed. in 2 vols.: Mathematical Biology: I. An Introduction, 2002 ISBN 0-387-95223-3; Mathematical Biology: II. Spatial Models and Biomedical Applications, 2003 ISBN 0-387-95228-4.
- E. Renshaw, Modelling biological populations in space and time. C.U.P., 1991. ISBN 0-521-44855-7
- S.I. Rubinow, Introduction to mathematical biology. John Wiley, 1975. ISBN 0-471-74446-8
- L.A. Segel, Modeling dynamic phenomena in molecular and cellular biology. C.U.P., 1984. ISBN 0-521-27477-X
- L. Preziosi, Cancer Modelling and Simulation. Chapman Hall/CRC Press, 2003. ISBN 1-58488-361-8.
- Theoretical biology
- Bertalanffy, L. v. 1932. Theoretische Biologie. Band I: Allgemeine Theorie, Physikochemie, Aufbau und Entwicklung des Organismus. Berlin: Gebrüder Borntraeger.
- Bonner, J. T. 1988. The Evolution of Complexity by Means of Natural Selection. Princeton: Princeton University Press.
- Hertel, H. 1963. Structure, Form, Movement. New York: Reinhold Publishing Corp.
- Mangel, M. 1990. Special Issue, Classics of Theoretical Biology (part 1). Bull. Math. Biol. 52(1/2): 1-318.
- Mangel, M. 2006. The Theoretical Biologist's Toolbox. Quantitative Methods for Ecology and Evolutionary Biology. Cambridge University Press.
- Prusinkiewicz, P. & Lindenmeyer, A. 1990. The Algorithmic Beauty of Plants. Berlin: Springer-Verlag.
- Reinke, J. 1901. Einleitung in die theoretische Biologie. Berlin: Verlag von Gebrüder Paetel.
- Schaxel, J. 1919. Grundzüge der Theorienbildung in der Biologie. Jena: Fischer.
- Thompson, D.W. 1942. On Growth and Form. 2nd ed. Cambridge: Cambridge University Press: 2. vols.
- Uexküll, J.v. 1920. Theoretische Biologie. Berlin: Gebr. Paetel.
- Vogel, S. 1988. Life's Devices: The Physical World of Animals and Plants. Princeton: Princeton University Press.
- Waddington, C.H. 1968-1972. Towards a Theoretical Biology. 4 vols. Edinburg: Edinburg University Press.
Further reading
- Hoppensteadt, F. (September 1995). "Getting Started in Mathematical Biology" (PDF). Notices of the American Mathematical Society..
- Reed, M. C. (March 2004). "Why Is Mathematical Biology So Hard?" (PDF). Notices of the American Mathematical Society..
- May, R. M. (2004). "Uses and Abuses of Mathematics in Biology". Science. 303 (5659): 790–793. doi:10.1126/science.1094442. PMID 14764866..
- Murray, J. D. (1988). "How the leopard gets its spots?". Scientific American. 258 (3): 80–87. doi:10.1038/scientificamerican0388-80..
- Schnell, S.; Grima, R.; Maini, P. K. (2007). "Multiscale Modeling in Biology" (PDF). American Scientist. 95: 134–142. doi:10.1511/2007.64.134..
- Chen, Katherine C.; Calzone, Laurence; Csikasz-Nagy, Attila; Cross, FR; Cross, Frederick R.; Novak, Bela; Tyson, John J. (2004). "Integrative analysis of cell cycle control in budding yeast". Mol Biol Cell. 15 (8): 3841–3862. doi:10.1091/mbc.E03-11-0794. PMC 491841
 . PMID 15169868..
. PMID 15169868.. - Csikász-Nagy, Attila; Battogtokh, Dorjsuren; Chen, Katherine C.; Novák, Béla; Tyson, John J. (2006). "Analysis of a generic model of eukaryotic cell-cycle regulation". Biophys J. 90 (12): 4361–4379. doi:10.1529/biophysj.106.081240. PMC 1471857
 . PMID 16581849..
. PMID 16581849.. - Fuss, H.; Dubitzky, Werner; Downes, C. Stephen; Kurth, Mary Jo (2005). "Mathematical models of cell cycle regulation". Brief Bioinform. 6 (2): 163–177. doi:10.1093/bib/6.2.163. PMID 15975225..
- Lovrics, Anna; Csikász-Nagy, Attila; Zsély1, István Gy; Zádor, Judit; Turányi, Tamás; Novák, Béla (2006). "Time scale and dimension analysis of a budding yeast cell cycle model". BMC Bioinform. 9 (7): 494. doi:10.1186/1471-2105-7-494..
Important names
External links
- The Society for Mathematical Biology
- Theoretical and mathematical biology website
- Konrad Lorenz Institute for Evolution and Cognition Research
- Complexity Discussion Group
- UCLA Biocybernetics Laboratory
- TUCS Computational Biomodelling Laboratory
- Nagoya University Division of Biomodeling
- Technische Universiteit Biomodeling and Informatics
- BioCybernetics Wiki, a vertical wiki on biomedical cybernetics and systems biology
- Bulletin of Mathematical Biology
- European Society for Mathematical and Theoretical Biology
- Journal of Mathematical Biology
- Biomathematics Research Centre at University of Canterbury
- Centre for Mathematical Biology at Oxford University
- Mathematical Biology at the National Institute for Medical Research
- Institute for Medical BioMathematics
- Mathematical Biology Systems of Differential Equations from EqWorld: The World of Mathematical Equations
- Systems Biology Workbench - a set of tools for modelling biochemical networks
- The Collection of Biostatistics Research Archive
- Statistical Applications in Genetics and Molecular Biology
- The International Journal of Biostatistics
- Theoretical Modeling of Cellular Physiology at Ecole Normale Superieure, Paris
- Biomathematics in the Philippines
- BioMath Modeler
- Lists of references
- A general list of Theoretical biology/Mathematical biology references, including an updated list of actively contributing authors.
- A list of references for applications of category theory in relational biology.
- An updated list of publications of theoretical biologist Robert Rosen
- Theory of Biological Anthropology (Documents No. 9 and 10 in English)
- Drawing the Line Between Theoretical and Basic Biology, a forum article by Isidro A. T. Savillo
- Mystery of Prime-numbered Cicadas
Related journals
- Acta Biotheoretica
- Bioinformatics
- Biological Theory
- BioSystems
- Bulletin of Mathematical Biology
- Ecological Modelling
- Journal of Mathematical Biology
- Journal of Theoretical Biology
- Journal of the Royal Society Interface
- Mathematical Biosciences
- Mathematical Biosciences and Engineering
- Medical Hypotheses
- Rivista di Biologia-Biology Forum
- Theoretical and Applied Genetics
- Theoretical Biology and Medical Modelling
- Theoretical Population Biology
- Theory in Biosciences (formerly: Biologisches Zentralblatt)