Phagocytosis
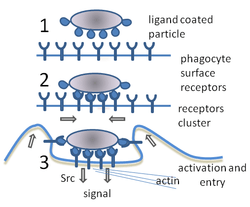
- Unbound phagocyte surface receptors do not trigger phagocytosis.
- Binding of receptors causes them to cluster.
- Phagocytosis is triggered and the particle is taken up by the phagocyte.
In cell biology, phagocytosis (from Ancient Greek φαγεῖν (phagein) , meaning "to devour", κύτος, (kytos) , meaning "cell", and -osis, meaning "process") is the process by which a cell—often a phagocyte or a protist—engulfs a solid particle to form an internal compartment known as a phagosome. It is distinct from other forms of endocytosis like (pinocytosis) that involves the internalization of extracellular liquids. Phagocytosis is involved in the acquisition of nutrients for some cells. The process is homologous to eating at the level of single-celled organisms; in multicellular animals, the process has been adapted to eliminate debris and pathogens, as opposed to taking in fuel for cellular processes, except in the case of the animal Trichoplax.
In an organism's immune system, phagocytosis is a major mechanism used to remove pathogens and cell debris. For example, when a macrophage ingests a pathogenic microorganism, the pathogen becomes trapped in a phagosome which then fuses with a lysosome to form a phagolysosome. Within the phagolysosome, enzymes and toxic peroxides digest the pathogen. Bacteria, dead tissue cells, and small mineral particles are all examples of objects that may be phagocytized.
History
Phagocytosis was first noted by Canadian physician William Osler (1876),[1] and later studied and named by Élie Metchnikoff (1880, 1883).
In immune system

Phagocytosis in mammalian immune cells is activated by attachment to pathogen-associated molecular patterns (PAMPS), which leads to NF-κB activation. Opsonins such as C3b and antibodies can act as attachment sites and aid phagocytosis of pathogens.[2]

- A particle is ingested by a phagocyte after antigens are recognized which results in the formation of a phagosome.
- The fusion of lysosomes with the phagosome creates a phagolysosome.
- The particle is broken down by the digestive enzymes found in the lysosomes. The resulting waste material is discharged from the phagocyte by exocytosis.
Engulfment of material is facilitated by the actin-myosin contractile system. The phagosome of ingested material is then fused with the lysosome, forming a phagolysosome and leading to degradation.
Degradation can be oxygen-dependent or oxygen-independent.
- Oxygen-dependent degradation depends on NADPH and the production of reactive oxygen species. Hydrogen peroxide and myeloperoxidase activate a halogenating system, which leads to the creation of hypochlorite and the destruction of bacteria.[3]
- Oxygen-independent degradation depends on the release of granules, containing proteolytic enzymes such as defensins, lysozyme, and cationic proteins. Other antimicrobial peptides are present in these granules, including lactoferrin, which sequesters iron to provide unfavourable growth conditions for bacteria.
It is possible for cells other than dedicated phagocytes (such as dendritic cells) to engage in phagocytosis. some of white blood cells in human immune system perform phagocytosis by gulping in some pathogenic and disease causing cells.[4]
In apoptosis
Following apoptosis, the dying cells need to be taken up into the surrounding tissues by macrophages in a process called efferocytosis. One of the features of an apoptotic cell is the presentation of a variety of intracellular molecules on the cell surface, such as calreticulin, phosphatidylserine (From the inner layer of the plasma membrane), annexin A1, oxidised LDL and altered glycans.[5] These molecules are recognised by receptors on the cell surface of the macrophage such as the phosphatidylserine receptor or by soluble (free-floating) receptors such as thrombospondin 1, GAS6, and MFGE8, which themselves then bind to other receptors on the macrophage such as CD36 and alpha-v beta-3 integrin. Defects in apoptotic cell clearance is usually associated with impaired phagocytosis of macrophages. Accumulation of apoptotic cell remnants often causes autoimmune disorders; thus pharmacological potentiation of phagocytosis has a medical potential in treatment of certain forms of autoimmune disorders.[6][7][8][9]
In protists
In many protists, phagocytosis is used as a means of feeding, providing part or all of their nourishment. This is called phagotrophic nutrition, distinguished from osmotrophic nutrition which takes place by absorption.
- In some, such as amoeba, phagocytosis takes place by surrounding the target object with pseudopods, as in animal phagocytes. In humans, entamoeba histolytica can phagocytose red blood cells. Entamoeba histolytica is an anaerobic parasitic protozoan, part of the genus Entamoeba. Predominantly infecting humans and other primates, E. histolytica is estimated to infect about 50 million people worldwide. Previously, it was thought that 10% of the world population was infected, but these figures predate the recognition that at least 90% of these infections were due to a second species, E. dispar. Mammals such as dogs and cats can become infected transiently, but are not thought to contribute significantly to transmission.The word histolytic literally means "Tissue destroyer".[10] This process is known as "erythrophagocytosis", and is considered the only reliable way to distinguish Entamoeba histolytica from noninvasive species such as Entamoeba dispar.[11]
- Ciliates also engage in phagocytosis.[12] In ciliates there is a specialized groove or chamber in the cell where phagocytosis takes place, called the cytostome or mouth.
As in phagocytic immune cells, the resulting phagosome may be merged with lysosomes containing digestive enzymes, forming a phagolysosome. The food particles will then be digested, and the released nutrients are diffused or transported into the cytosol for use in other metabolic processes.
Mixotrophy can involve phagotrophic nutrition and phototrophic nutrition.[13]
See also
- Active transport
- Antigen presentation
- Antigen presenting cell
- Emperipolesis
- Endosymbionts in protists
- Paracytophagy
- Phagoptosis
- Residual body
- Cell wall
References
- ↑ "The Osler slide, a demonstration of phagocytosis from 1876: Reports of phagocytosis before Metchnikoff's 1880 paper". Cellular Immunology. 240: 1–4. doi:10.1016/j.cellimm.2006.05.008.
- ↑ The Immune System, Peter Parham, Garland Science, 2nd edition
- ↑ http://www.colorado.edu/intphys/iphy3700/vitCHemila92.pdf
- ↑ Ishimoto, H; Yanagihara, K; Araki, N; Mukae, H; Sakamoto, N; Izumikawa, K; Seki, M; Miyazaki, Y; Hirakata, Y; Mizuta, Y; Yasuda, K; Kohno, S (July 2008). "Single-cell observation of phagocytosis by human blood dendritic cells". Jpn. J. Infect. Dis. 61 (4): 294–7. PMID 18653972.
- ↑ Bilyy, R; Shkandina, T; Tomin, A; Muñoz, LE; Franz, S; Antonyuk, V; Kit, YY; Zirngibl, M; Fürnrohr, BG; Janko, C; Lauber, K; Schiller, M; Schett, G; Stoika, RS; Herrmann, M (January 2012). "Macrophages discriminate glycosylation patterns of apoptotic cell-derived microparticles". J Biol Chem. 287 (1): 496–503. doi:10.1074/jbc.M111.273144. PMC 3249103
 . PMID 22074924.
. PMID 22074924. - ↑ Mukundan, Lata; Odegaard, Justin I; Morel, Christine R; Heredia, Jose E; Mwangi, Julia W; Ricardo-Gonzalez, Roberto R; Goh, Y P Sharon; Eagle, Alex Red; Dunn, Shannon E; Awakuni, Jennifer U H; Nguyen, Khoa D; Steinman, Lawrence; Michie, Sara A; Chawla, Ajay (18 October 2009). "PPAR-δ senses and orchestrates clearance of apoptotic cells to promote tolerance". Nature Medicine. 15 (11): 1266–1272. doi:10.1038/nm.2048. PMC 2783696
 . PMID 19838202.
. PMID 19838202. - ↑ Roszer, T; Menéndez-Gutiérrez, MP; Lefterova, MI; Alameda, D; Núñez, V; Lazar, MA; Fischer, T; Ricote, M (Jan 1, 2011). "Autoimmune kidney disease and impaired engulfment of apoptotic cells in mice with macrophage peroxisome proliferator-activated receptor gamma or retinoid X receptor alpha deficiency". Journal of immunology (Baltimore, Md. : 1950). 186 (1): 621–31. doi:10.4049/jimmunol.1002230. PMID 21135166.
- ↑ Kruse, K; Janko, C; Urbonaviciute, V; Mierke, CT; Winkler, TH; Voll, RE; Schett, G; Muñoz, LE; Herrmann, M (September 2010). "Inefficient clearance of dying cells in patients with SLE: anti-dsDNA autoantibodies, MFG-E8, HMGB-1 and other players". Apoptosis. 15 (9): 1098–113. doi:10.1007/s10495-010-0478-8. PMID 20198437.
- ↑ Han, CZ; Ravichandran, KS (Dec 23, 2011). "Metabolic connections during apoptotic cell engulfment". Cell. 147 (7): 1442–5. doi:10.1016/j.cell.2011.12.006. PMC 3254670
 . PMID 22196723.
. PMID 22196723. - ↑ Boettner DR; Huston CD; Linford AS; et al. (January 2008). "Entamoeba histolytica phagocytosis of human erythrocytes involves PATMK, a member of the transmembrane kinase family". PLoS Pathog. 4 (1): e8. doi:10.1371/journal.ppat.0040008. PMC 2211552
 . PMID 18208324.
. PMID 18208324. - ↑ "DPDx — Amebiasis". Retrieved 2008-12-30.
- ↑ Grønlien HK, Berg T, Løvlie AM; Berg; Løvlie (July 2002). "In the polymorphic ciliate Tetrahymena vorax, the non-selective phagocytosis seen in microstomes changes to a highly selective process in macrostomes". J. Exp. Biol. 205 (Pt 14): 2089–97. PMID 12089212.
- ↑ Stibor H, Sommer U; Sommer (April 2003). "Mixotrophy of a photosynthetic flagellate viewed from an optimal foraging perspective". Protist. 154 (1): 91–8. doi:10.1078/143446103764928512. PMID 12812372.
External links
- Phagocytosis at the US National Library of Medicine Medical Subject Headings (MeSH)