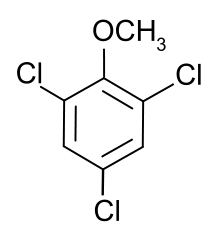
2,4,6-Trichloroanisole
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,3,5-Trichloro-2-methoxybenzene | |
| Other names
2,4,6-Trichloroanisole TCA 2,4,6-Trichloromethoxybenzene | |
| Identifiers | |
| 87-40-1 | |
| 3D model (Jmol) | Interactive image Interactive image |
| ChEBI | CHEBI:19333 |
| ChemSpider | 6620 |
| ECHA InfoCard | 100.001.585 |
| KEGG | C11510 |
| RTECS number | MFCD00000588 |
| |
| |
| Properties | |
| C7H5Cl3O | |
| Molar mass | 211.47 g·mol−1 |
| Melting point | 60 to 62 °C (140 to 144 °F; 333 to 335 K) |
| Boiling point | 140 °C (284 °F; 413 K) at 28 torr |
| Hazards | |
| R-phrases | R22 R36 |
| S-phrases | S26 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
2,4,6-Trichloroanisole (TCA) is a chemical compound that is a chlorinated derivative of anisole. TCA is a fungal metabolite of 2,4,6-trichlorophenol, which is used as a fungicide. It can be found in minute traces on packaging materials stored in the presence of fiberboard treated with trichlorophenol.
TCA is the chemical primarily responsible for cork taint in wines. TCA has also been implicated as a major component of the "Rio defect" in coffees from Central and South America, which refers to a taste described as medicinal, phenolic, or iodine-like.[1]
TCA is usually produced when naturally occurring airborne fungi and bacteria (usually Aspergillus sp., Penicillium sp., Actinomycetes, Botrytis cinerea, Rhizobium sp., or Streptomyces) are presented with chlorinated phenolic compounds, which they then convert into chlorinated anisole derivatives. The chlorophenols can originate from various contaminants such as those found in some pesticides and wood preservatives. Chlorophenols can also be a product of the chlorine bleaching process used to sterilize or bleach wood, paper, and other materials; they can be synthesized by reaction of hypochlorites with lignin. They can also migrate from other objects such as shipping pallets treated by chlorophenols.
The odor of TCA is not directly perceived. Instead, the molecule distorts the perception of smell by suppressing olfactory signal transduction.[2] The effect occurs at very low concentrations (single parts per trillion), so even very minute amounts of TCA can be detected. It causes unpleasant earthy, musty and moldy aromas.
See also
References
- ↑ Spadone, Jean Claude; Jean Claude Spadone; Gary Takeoka; Remy Liardon (1990). "Analytical investigation of Rio off-flavor in green coffee". Journal of Agricultural and Food Chemistry. 38: 226–233. doi:10.1021/jf00091a050.
- ↑ Takeuchi, Hiroko; Hiroyuki Kato; Takashi Kurahashi (2013-09-16). "2,4,6-Trichloroanisole is a potent suppressor of olfactory signal transduction". Proceedings of the National Academy of Sciences: 201300764. doi:10.1073/pnas.1300764110. ISSN 1091-6490. Retrieved 2013-09-17.